 
Carbon tetrachloride, ethyl alcohol, methyl alcohol, acetone, acetic anhydride.Point A: streams leaving stage 2 (L2, V2) Point B: vapor stream leaving stage 5 (V5) liquid stream leaving stage 4 (L4) Temp. Of stage 2: know K y 2 / x 2, can get T from temperature-composition graph or DePriester chart of K f(T,p). In reboiler: same as above (reboiler is an equilibrium stage.) 4A2. DePriester charts in other temperature and pressure units are given by Perry and Green (1997), Perry et al.A.Depending on the system under study, any one of several approaches may be used to determine K-values. 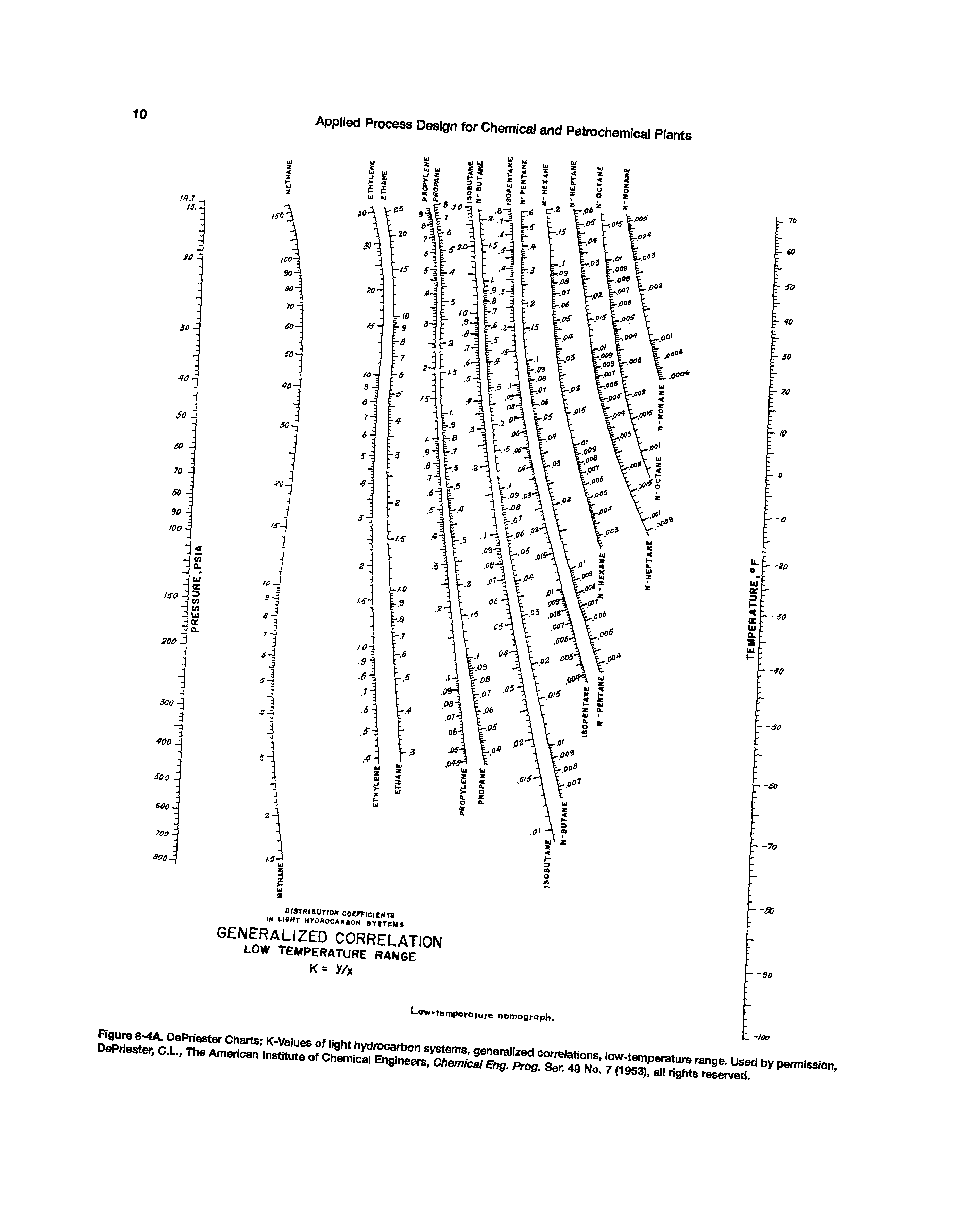
The DePriester charts have been fit to the following equation (McWilliams, 1973): ln K aT1/ T2 + aT2 / T + aT6 + ap1 ln p + ap2 /p2 + ap3 /p (2-30) Note that T is in º R and p is in psia in Eq. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
